When an experiment fails, have you ever pointed the finger at expensive reagents or complex instruments? But sometimes, the real "culprit" may be quietly hiding in the corner - that seemingly ordinary bottle of buffer reagent. Although it is inconspicuous, it is the lifeline for maintaining the accuracy of the experiment. Today, we will uncover the secret of this "invisible guardian".
Ⅰ. Buffer: What is it? Why is it indispensable in the laboratory?
Simply put, a buffer is a special solution system that can resist the effects of a small amount of added acid, alkali or dilution and maintain a relatively stable pH value of the solution. It is usually composed of a pair of "partners" - a weak acid and its conjugate base (or a weak base and its conjugate acid), such as acetic acid/sodium acetate (HAc/Ac-), sodium dihydrogen phosphate/disodium hydrogen phosphate (NaH₂PO₄/Na₂HPO₄), etc., which are common in laboratories.
Core ability: Stabilize pH and resist changes!
When a small amount of acidic or alkaline substances are accidentally introduced into the experiment, the "partner" in the buffer will react quickly, like an efficient "hydrogen ion gatekeeper", absorbing or releasing it, thereby avoiding drastic fluctuations in the pH value of the solution. This ability is essential for many biochemical reactions that rely on a specific pH environment.
Ⅱ. Laboratory stage: an indispensable protagonist and supporting role
1. "Life support system" of biochemical experiments:
Enzyme activity research: Most enzymes have optimal activity only within a narrow pH range. Buffers (such as Tris-HCl, PBS) provide a stable "workbench" for enzyme reactions to ensure reliable results.
Protein purification and analysis: From cell lysis, chromatographic separation (ion exchange, affinity chromatography) to electrophoresis (such as SDS-PAGE), protein crystallization and storage, each step requires a buffer with a specific pH to maintain the structure, charge and solubility of the protein and prevent denaturation or precipitation.
Nucleic acid manipulation: PCR reaction, DNA/RNA extraction, enzyme digestion, ligation, hybridization and other processes are extremely sensitive to pH. Commonly used buffers such as TE (Tris-EDTA) can stabilize the structure of nucleic acids and protect them from nuclease degradation.
2. "Lullaby" of cell culture: Cell culture medium (such as DMEM, RPMI) itself is a complex buffer system (often relying on sodium bicarbonate/carbon dioxide system). Adding additional buffers (such as HEPES) can provide cells with a more stable pH environment, especially when changing liquids, observing or when carbon dioxide concentrations may fluctuate, to ensure healthy cell growth.
3. The "stabilizing force" of analytical chemistry:
Chromatographic separation: In high performance liquid chromatography (HPLC) and ion chromatography (IC), the pH of the mobile phase is the key control parameter for the separation effect, and buffers (such as phosphates and acetates) ensure the reproducibility of the separation process.
Titration analysis: Some titration reactions need to be carried out at a constant pH or indicate the end point when a specific pH is reached, and the buffer plays a stabilizing role.
Preparation of standard solutions: The pH value of many standard solutions needs to be precisely controlled, and buffers are the basis.
Ⅲ. Selection and use: a lot of knowledge
Choose the right buffer system:
pKa value is key: The effective buffering range of a buffer is usually within the range of its pKa ± 1 value. Be sure to choose a buffer with a pKa value close to the working pH you need! For example, phosphate (pKa ~ 7.2) is often used in the neutral pH range, and Tris (pKa ~ 8.1) is often used in alkaline.
Compatibility: Consider whether the buffer components interfere with your experimental reaction (e.g. phosphate may interfere with some phosphorylation studies, and borate may react with sugars).
Ionic strength and temperature: Ionic strength affects enzyme activity, solubility, etc.; temperature changes can significantly affect the pKa value of buffers such as Tris (Tris has a large pKa temperature coefficient of about -0.031/℃).

Ⅳ. List of Common Buffers
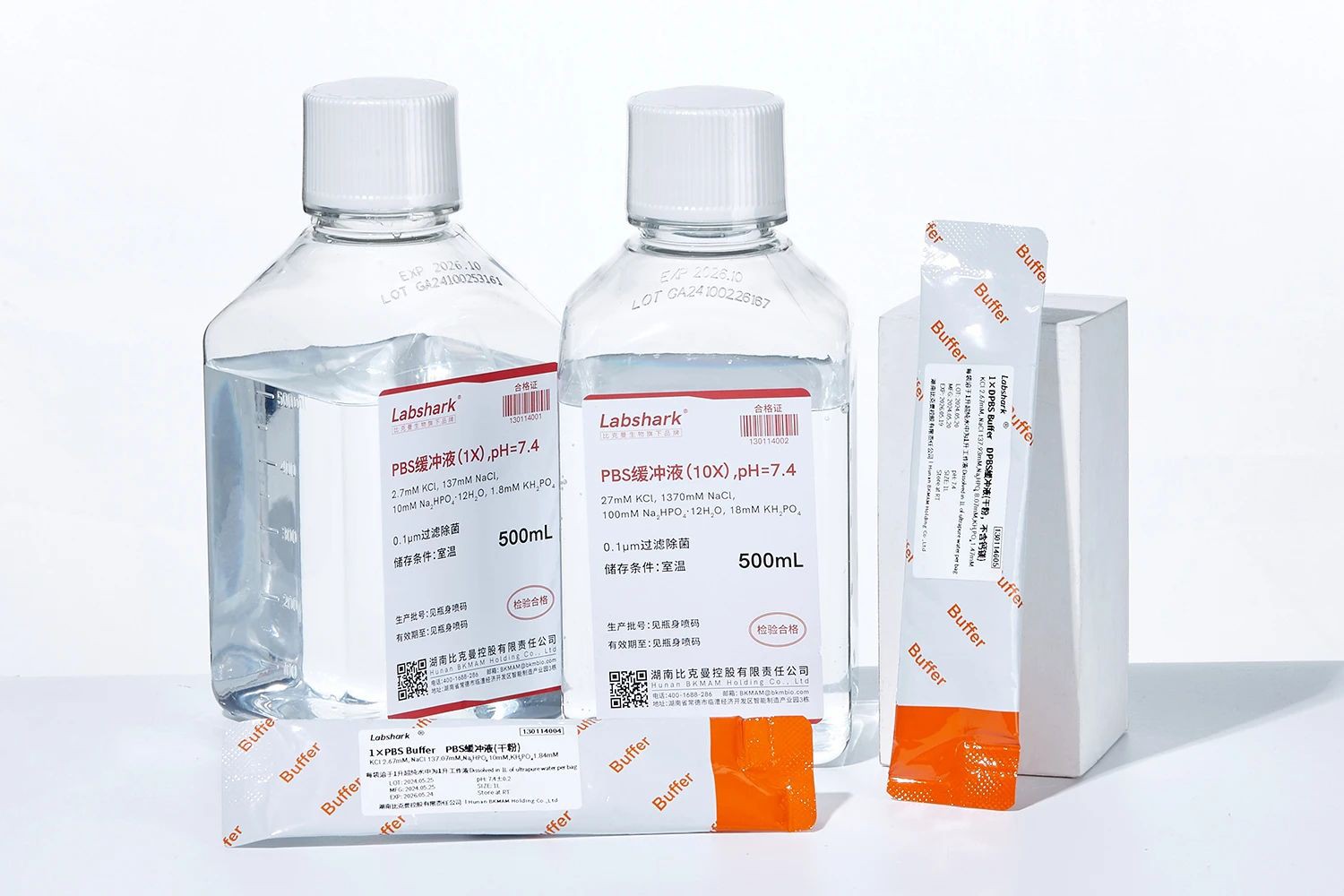
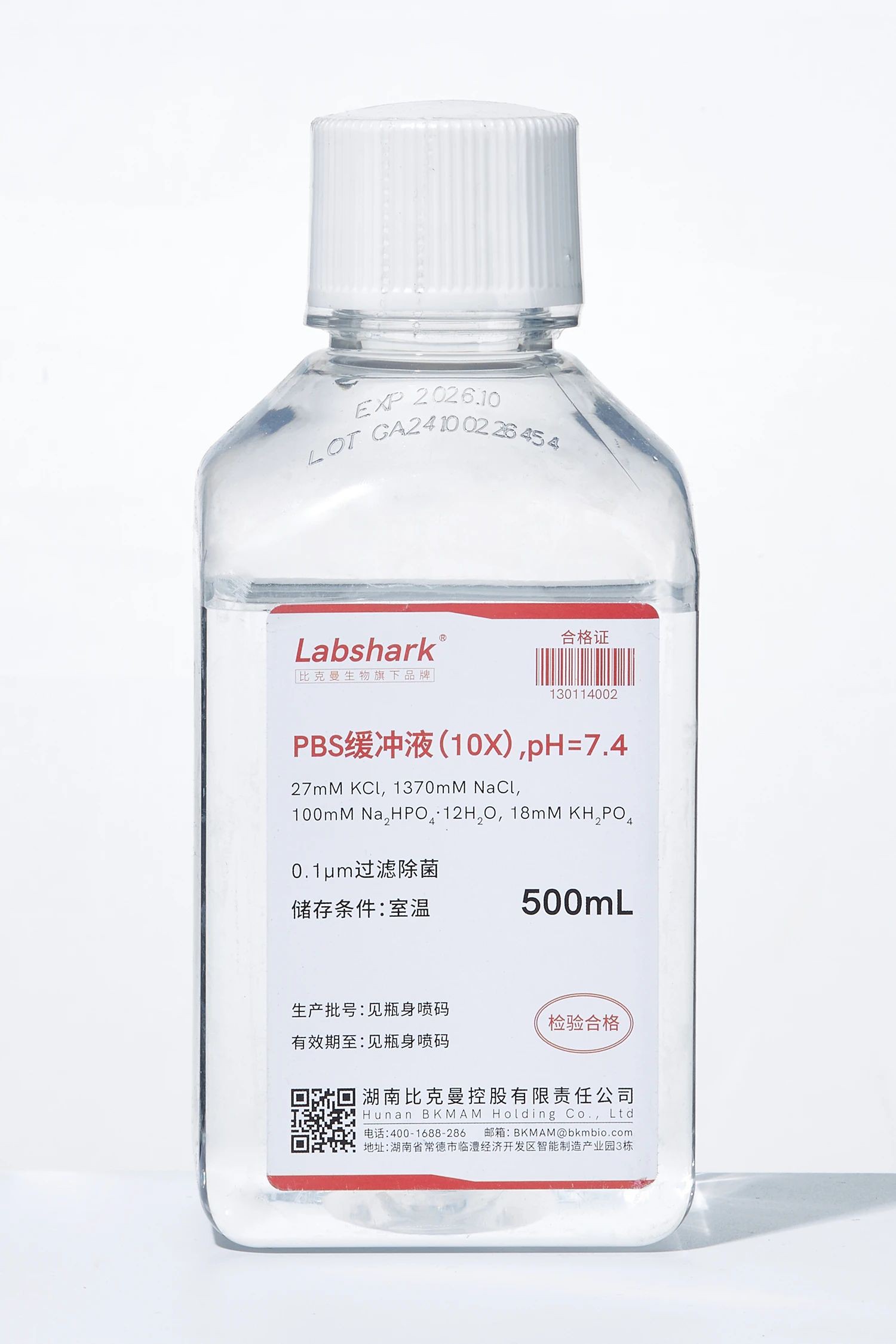
1. PBS buffer series
PBS buffer 1× (pH7.4),PBS buffer 10× (pH7.4), PBS phosphate powder (without calcium and magnesium ions): washing cells in cell culture experiments (such as before changing the culture medium); diluting antibodies and washing ELISA plates in immunology experiments; maintaining osmotic pressure and pH stability in protein/nucleic acid experiments, etc.
DPBS buffer (without calcium and magnesium), DPBS phosphate powder (without calcium and magnesium ions): flow cytometry in cell experiments, washing after cell digestion (to avoid calcium and magnesium interfering with enzyme activity); diluting enzymes or conducting ion-sensitive experiments in molecular biology (such as certain PCR reactions), etc.
PBST buffer (1×), PBST buffer (10×): used for blocking and washing steps in Western Blot or immunohistochemistry, reducing nonspecific binding, etc.
2. TBS buffer series
TBS buffer (1×), TBS buffer (10×): replace PBS buffer in immunology experiments (to avoid phosphate interference with certain antibody binding); stabilize alkaline pH environment in protein experiments, etc.
3. HBSS buffer series
HBSS buffer (calcium and magnesium free, phenol red free)/Hank's buffer (calcium and magnesium free): cell separation and centrifugal washing in cell experiments (such as flow cytometry); short-term cell maintenance (such as live cell imaging), etc. HBSS buffer (calcium and magnesium free, phenol red free): maintain cell metabolic activity in cell culture (calcium and magnesium participate in signal transduction); cell migration/invasion experiments.
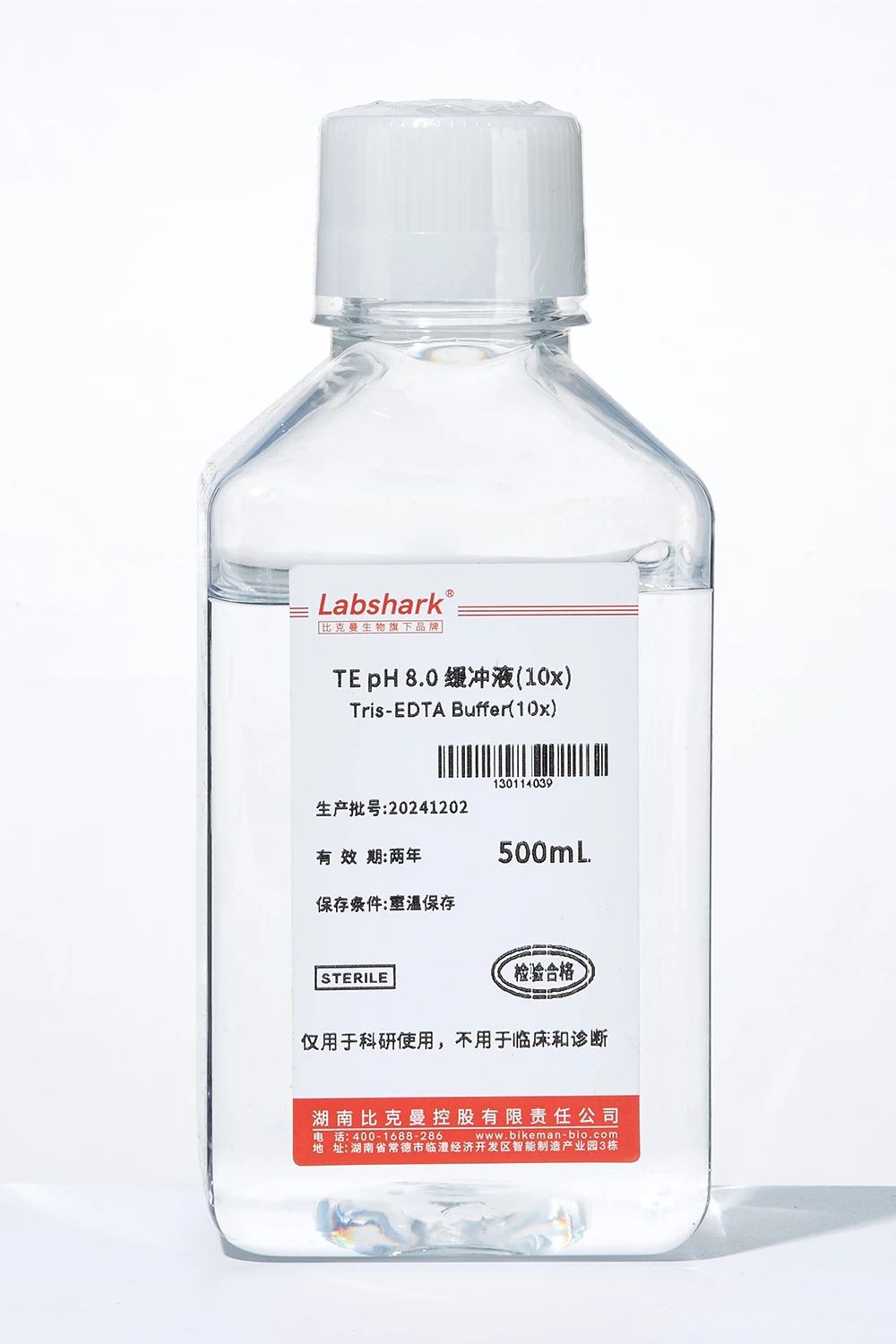
4. TE buffer series
TE buffer (1×), TE buffer (10×): dissolve and store DNA/RNA (Tris maintains pH, EDTA chelates metal ions to inhibit nuclease activity); dilute DNA for PCR, sequencing and other experiments.
5. Electrophoresis buffer series
TAE buffer (1×): conventional DNA agarose gel electrophoresis (low resolution, suitable for large DNA fragments). TBE buffer (1×), TBE buffer (10×): high-resolution DNA/RNA electrophoresis (such as small fragments or polyacrylamide gel electrophoresis); long-term electrophoresis (buffering capacity is better than TAE).
6. Western Blot Experiment Buffer Series
Western Blot Transfer Buffer (10×): Buffer used to transfer proteins from gel to membrane (such as PVDF membrane), containing Tris, glycine and methanol.
Tris-HCl buffer (1mol/L, pH6.8): Preparation of SDS-PAGE concentrated gel, etc.
Tris-HCl buffer (1.5mol/L, pH8.8): Preparation of SDS-PAGE separation gel, etc.
7. Standard pH buffer series
pH=4.00/6.86/9.18 buffer (prepared 250mL): standard solution for calibrating pH meter (e.g. pH6.86 is neutral standard, pH4.00 and 9.18 are acidic and alkaline standards).
Buffer reagents are by no means a basic supporting role in the laboratory. Their core value lies in providing a precise and stable proton environment, which is the cornerstone for maintaining the thermodynamic equilibrium and kinetic rate of most biochemical reactions. From the microenvironment regulation of enzyme active sites, to the guarantee of conformational stability of biomacromolecules, to the maintenance of transmembrane ion gradients, the pH buffering capacity of the buffer system is an indispensable prerequisite for the repeatability and reliability of experimental data. Beekman will escort you on your scientific research journey.
Hunan BKMAM Holding Co., Ltd
Website:www.bkmbio.com
Email:info@bkmbio.com





